Astronomy 210 Quiz 4, spring semester 2015
Cuesta College, San Luis Obispo, CA
Compared to the oldest surfaces on Mercury, the more recently developed surfaces have __________ craters.
(A) fewer.
(B) volcanic.
(C) darker.
(D) collapsed.
Correct answer (highlight to unhide): (A)
Because of the rate of bombardment in the solar system has decreased over time, the density of impact craters indicates the surface age of a terrestrial planet, where a heavily cratered region is relatively older than a region with fewer craters, which would have experienced more recent geological activity that would remove/cover craters.
Section 36074
Exam code: quiz04NoR3
(A) : 26 students
(B) : 5 students
(C) : 4 students
(D) : 3 students
Success level: 71% (including partial credit for multiple-choice)
Discrimination index (Aubrecht & Aubrecht, 1983): 0.60
Section 30676
Exam code: quiz04s0U7
(A) : 37 students
(B) : 1 student
(C) : 3 students
(D) : 5 students
Success level: 80% (including partial credit for multiple-choice)
Discrimination index (Aubrecht & Aubrecht, 1983): 0.54
Showing posts with label solidification age. Show all posts
Showing posts with label solidification age. Show all posts
20150405
20130520
Astronomy quiz question: comparative surface dating of terrestrial planets
Astronomy 210 Quiz 7, spring semester 2013
Cuesta College, San Luis Obispo, CA
__________ are used to compare the age of different regions on terrestrial planets.
(A) Impact crater densities.
(B) Infrared measurements.
(C) Volcano heat maps.
(D) Polar ice caps.
Correct answer (highlight to unhide): (A)
Because of the rate of bombardment in the solar system has decreased over time, the density of impact craters indicates the surface age of a terrestrial planet, where a heavily cratered region is relatively older than a smoother region, which would have experience more recent geological activity.
Section 36074
Exam code: quiz07n0iC
(A) : 20 students
(B) : 3 students
(C) : 2 students
(D) : 1 student
Success level: 79% (including partial credit for multiple-choice)
Discrimination index (Aubrecht & Aubrecht, 1983): 0.57
Section 30676
Exam code: quiz07sEcn
(A) : 27 students
(B) : 9 students
(C) : 2 students
(D) : 4 students
(No response) : 1 student
Success level: 66% (including partial credit for multiple-choice)
Discrimination index (Aubrecht & Aubrecht, 1983): 0.54
Cuesta College, San Luis Obispo, CA
__________ are used to compare the age of different regions on terrestrial planets.
(A) Impact crater densities.
(B) Infrared measurements.
(C) Volcano heat maps.
(D) Polar ice caps.
Correct answer (highlight to unhide): (A)
Because of the rate of bombardment in the solar system has decreased over time, the density of impact craters indicates the surface age of a terrestrial planet, where a heavily cratered region is relatively older than a smoother region, which would have experience more recent geological activity.
Section 36074
Exam code: quiz07n0iC
(A) : 20 students
(B) : 3 students
(C) : 2 students
(D) : 1 student
Success level: 79% (including partial credit for multiple-choice)
Discrimination index (Aubrecht & Aubrecht, 1983): 0.57
Section 30676
Exam code: quiz07sEcn
(A) : 27 students
(B) : 9 students
(C) : 2 students
(D) : 4 students
(No response) : 1 student
Success level: 66% (including partial credit for multiple-choice)
Discrimination index (Aubrecht & Aubrecht, 1983): 0.54
20120514
Physics quiz question: radioactive decay age
Physics 205B Quiz 7, spring semester 2012
Cuesta College, San Luis Obispo, CA
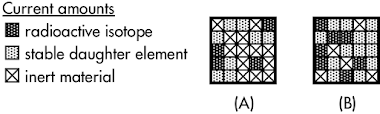 Two samples have certain amounts of a radioactive isotope, an embedded gaseous daughter element, and inert material (which is not involved in the decay process). As determined by radioactive dating, __________ has an older solidification age.
Two samples have certain amounts of a radioactive isotope, an embedded gaseous daughter element, and inert material (which is not involved in the decay process). As determined by radioactive dating, __________ has an older solidification age.
(A) sample A.
(B) sample B.
(C) (There is a tie.)
(D) (Cannot be determined.)
Correct answer (highlight to unhide): (A)
The solidification age of a sample (how long ago has it been since it was last molten) is determined by the ratio of decay products to its unstable isotopes. A larger ratio of decay products to unstable isotopes corresponds to a sample with a very old solidification age.
Sample A started with 12 unstable isotope squares when it first solidified (as any gaseous decay products would have been released when it was melted), of which 4 remain today, such that it has 33% of its original radioactivity today. Sample B started with 20 unstable isotope squares when it first solidified, of which 7 remain today, such that it has 35% of its original radioactivity today.
Section 30882
Exam code: quiz07b3Ta
(A) : 4 students
(B) : 1 student
(C) : 3 students
(D) : 0 students
Success level: 60%
Discrimination index (Aubrecht & Aubrecht, 1983): 0.46
Cuesta College, San Luis Obispo, CA
 Two samples have certain amounts of a radioactive isotope, an embedded gaseous daughter element, and inert material (which is not involved in the decay process). As determined by radioactive dating, __________ has an older solidification age.
Two samples have certain amounts of a radioactive isotope, an embedded gaseous daughter element, and inert material (which is not involved in the decay process). As determined by radioactive dating, __________ has an older solidification age.(A) sample A.
(B) sample B.
(C) (There is a tie.)
(D) (Cannot be determined.)
Correct answer (highlight to unhide): (A)
The solidification age of a sample (how long ago has it been since it was last molten) is determined by the ratio of decay products to its unstable isotopes. A larger ratio of decay products to unstable isotopes corresponds to a sample with a very old solidification age.
Sample A started with 12 unstable isotope squares when it first solidified (as any gaseous decay products would have been released when it was melted), of which 4 remain today, such that it has 33% of its original radioactivity today. Sample B started with 20 unstable isotope squares when it first solidified, of which 7 remain today, such that it has 35% of its original radioactivity today.
Section 30882
Exam code: quiz07b3Ta
(A) : 4 students
(B) : 1 student
(C) : 3 students
(D) : 0 students
Success level: 60%
Discrimination index (Aubrecht & Aubrecht, 1983): 0.46
20110113
Astronomy final exam question: Earth vs. moon rock sample ages
Astronomy 210 Final Exam, Fall Semester 2010
Cuesta College, San Luis Obispo, CA
[20 points.] Why do rock samples from the moon's surface have older radioactive dating ages than rock samples from the Earth's ocean bottoms? Explain by discussing properties of planets, and radioactive dating ages.
Solution and grading rubric:
Section 70160
p: 7 students
r: 12 students
t: 6 students
v: 10 students
x: 1 student
y: 1 student
z: 1 student
A sample "p" response (from student 1111):
Another sample "p" response (from student 1104):
Yet another sample "p" response (from student 0307):
A sample "v" response (from student 0001), discussing wear and tear on the moon:
A sample "v" response (from student 1919), discussing the fission theory of the formation of the moon:
A sample "v" response (from student 8008), discussing the large impact theory of the formation of the moon:
Cuesta College, San Luis Obispo, CA
[20 points.] Why do rock samples from the moon's surface have older radioactive dating ages than rock samples from the Earth's ocean bottoms? Explain by discussing properties of planets, and radioactive dating ages.
Solution and grading rubric:
- p = 20/20:
Correct. Primarily understands renewal versus static nature of Earth's and the moon's surfaces, with at least some discussion of how radioactive dating is "reset" by melting. - r = 16/20:
Nearly correct (explanation weak, unclear or only nearly complete); includes extraneous/tangential information; or has minor errors. Radioactive dating "reset" discussion is missing or only implied. - t = 12/20:
Contains right ideas, but discussion is unclear/incomplete or contains major errors. At least recognizes the relative activities of Earth's and the moon's surfaces, but may involve volcanism rather than plate tectonics. - v = 8/20:
Limited relevant discussion of supporting evidence of at least some merit, but in an inconsistent or unclear manner. Discussion based on "wear and tear," "exposure," or moon formation theories. - x = 4/20:
Implementation/application of ideas, but credit given for effort rather than merit.
y = 2/20: Irrelevant discussion/effectively blank. - y = 2/20:
Irrelevant discussion/effectively blank. - z = 0/20:
Blank.
Section 70160
p: 7 students
r: 12 students
t: 6 students
v: 10 students
x: 1 student
y: 1 student
z: 1 student
A sample "p" response (from student 1111):

Another sample "p" response (from student 1104):

Yet another sample "p" response (from student 0307):

A sample "v" response (from student 0001), discussing wear and tear on the moon:

A sample "v" response (from student 1919), discussing the fission theory of the formation of the moon:

A sample "v" response (from student 8008), discussing the large impact theory of the formation of the moon:

20080320
Astronomy midterm question: solidification age reset
Astronomy 10 Midterm 2, spring semester 2008
Cuesta College, San Luis Obispo, CA
Astronomy 10 learning goal Q4.2
A rock sample with an extremely old solidification age (as determined by radioactive dating) is heated until it is molten, and then cooled back down to a solid. Discuss what happens to its solidification age, and explain why this happens.
Solution and grading rubric:
Section 4160
p: 2 students
r: 2 students
t: 6 students
v: 26 students
x: 2 students
y: 2 students
z: 0 students
Section 5166
p: 5 students
r: 15 students
t: 13 students
v: 28 students
x: 2 students
y: 0 students
z: 1 student
A sample "p" response (from student 2501):
A sample "x" response (from student 3635), with credit given for effort, rather than merit.
Previous post: Astronomy quiz question: bubble, bubble, toil and trouble
Cuesta College, San Luis Obispo, CA
Astronomy 10 learning goal Q4.2
A rock sample with an extremely old solidification age (as determined by radioactive dating) is heated until it is molten, and then cooled back down to a solid. Discuss what happens to its solidification age, and explain why this happens.
Solution and grading rubric:
- p:
Correct. The solidification age of a rock is determined by comparing the amount of gaseous decay products to the amount of unstable radioactive isotopes in the sample; a higher ratio of gaseous products to unstable isotopes corresponds to a sample that had solidified a long time ago. Heating the rock until molten would release the gaseous decay products, such that when it cools back down to a solid, it would have nothing to compare to the unstable isotopes that remain in the sample, effectively giving it a zero solidification age. - r:
Nearly correct (explanation weak, unclear or only nearly complete); includes extraneous/tangential information; or has minor errors. Recognizes that during melting, the amount of decay products in the sample somehow goes to zero, or is transmuted, thus "resetting" the solidification age to zero when it cools and solidifies. - t:
Contains right ideas, but discussion is unclear/incomplete or contains major errors. Solidification age is reset, but due to loss/gain/renewal of unstable isotopes. - v:
Limited relevant discussion of supporting evidence of at least some merit, but in an inconsistent or unclear manner. - x:
Implementation/application of ideas, but credit given for effort rather than merit. - y:
Irrelevant discussion/effectively blank. - z:
Blank.
Section 4160
p: 2 students
r: 2 students
t: 6 students
v: 26 students
x: 2 students
y: 2 students
z: 0 students
Section 5166
p: 5 students
r: 15 students
t: 13 students
v: 28 students
x: 2 students
y: 0 students
z: 1 student
A sample "p" response (from student 2501):

A sample "x" response (from student 3635), with credit given for effort, rather than merit.

Previous post: Astronomy quiz question: bubble, bubble, toil and trouble
20070301
Astronomy quiz question: bubble, bubble, toil and trouble
 Astronomy 10 Quiz 4, fall semester 2006
Astronomy 10 Quiz 4, fall semester 2006Cuesta College, San Luis Obispo, CA
Astronomy 10 learning goal Q4.2
Consider a sample with differing amounts of unstable isotopes, embedded gaseous decay products, and inert material (which is not involved in the radioactive decay process), schematically shown at right above. This sample is then melted completely, and then cooled and solidified. Which one of the following choices (A)-(E) schematically shown below best corresponds to the sample after it has resolidified? Clearly circle your answer.

Correct answer (highlight to unhide): (B)
The solidification age of a sample (how long ago has it been since it was last molten) is determined by the ratio of decay products to its unstable isotopes. A larger ratio of decay products to unstable isotopes corresponds to a sample with a very old solifidification age. Because these decay products are typically gaseous, when a sample is heated up to a molten state, the decay products bubble out, and thus the ratio of decay products to unstable isotopes is "zeroed out," corresponding to a zero-age solification age as it cools back down and resolidifies.
The plurality of (A) responses seems to indicate that students are thinking that heating up and melting a sample "resets" the decay products back into the original unstable isotopes.
Student responses:
(A): 11 students
(B): 7 students
(C): 5 students
(D): 4 students
(E): 6 students
Success level: 21%
Discrimination index (Aubrecht & Aubrecht, 1983): 0.86
Subscribe to:
Comments (Atom)